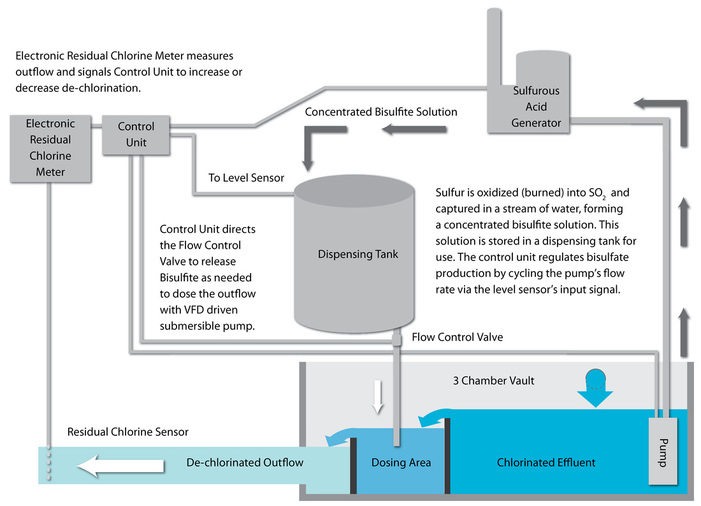
Dechlorination
Process
ERT dechlor is superior for the environment because it generates neutralizing bisulfite through the production of sulfurous acid and does not introduce sodium into the treated waste stream the way any of the sodium sulfite neutralization methods do; thereby avoiding the introduction of added salts into the environment. In most cases, ERT's dechlor application is at least 10% less expensive than the cost of dechlor using sodium bisulfite as a chlorine neutralization agent and its on-site generation from elemental raw sulfur is much safer than transport and use of gaseous SO2.
Advantages
ERT dechlor is superior for the environment because it generates neutralizing bisulfite through the production of sulfurous acid and does not introduce sodium into the treated waste stream the way any of the sodium sulfite neutralization methods do; thereby avoiding the introduction of added salts into the environment. In most cases, ERT's dechlor application is at least 10% less expensive than the cost of dechlor using sodium bisulfite as a chlorine neutralization agent and its on-site generation from elemental raw sulfur is much safer than transport and use of gaseous SO2.
Advantages
- Proven technology to neutralize free chlorine in effluent
- Improves water quality by reducing monovalent salts in discharge
- Employs highly evolved equipment with decades-long proven safety record generates bisulfate on site from raw sulfur
- Often much less expensive than other methods of chlorine neutralization
- Service is typically available with no required capital investment by customer. ERT also offers the necessary equipment for sale and in-house operation.
- Process utilizes on-site storage of safe prilled sulfur rather than hazardous chemicals